Atomic Models
- Describe the structure of an atom in terms of fundamental particles. Describe the proton, electron and neutron in terms of their mass and charge.
- Define the terms atomic mass, atomic number, mass number and isotope. Use and interpret symbols in the form AZX for elements
- Examine the development for ‘Models of the atom’, including Dalton and Thompson. Describe the operation of Rutherford’s “gold foil” experiment. Describe, with a diagram, the main features of the Rutherford model of the atom.
|
| ||||||||||||
Radioactivity and its uses
- Describe the three basic forms of radioactivity. Discuss their ionising power and penetration power. The effects of radiation and radiation monitoring.
- Balance nuclear equations. State the rules for balancing nuclearequations. Write balanced nuclear equations.
- Make calculations based upon the concept of half life. Understand the exponential nature of radioactive decay
- Outline some of the uses of radioactivity, eg carbon dating.
- Name the main sources of the radiation we receive.
- Explain what affects the amount of radiation we receive from the above sources.
- Outline some things which can be done to protect us from radiation.
- Name two devices used to detect radiation and describe briefly when they would be used.
| 3_radioactivity___equations.pptx | |
| File Size: | 738 kb |
| File Type: | pptx |
| 4_half_life___carbon_dating.pptx | |
| File Size: | 822 kb |
| File Type: | pptx |
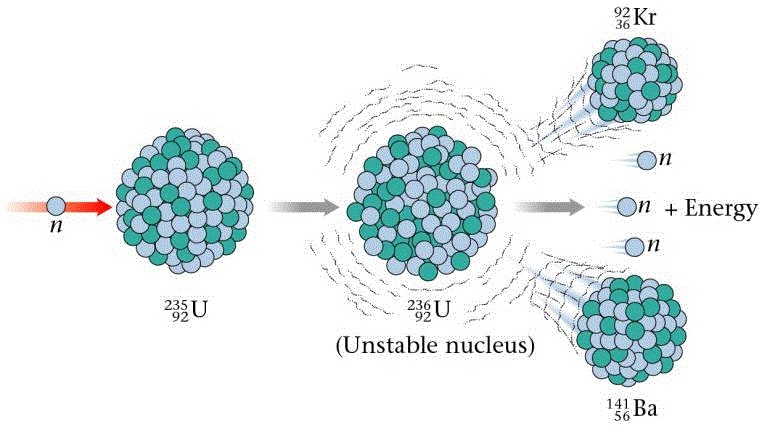
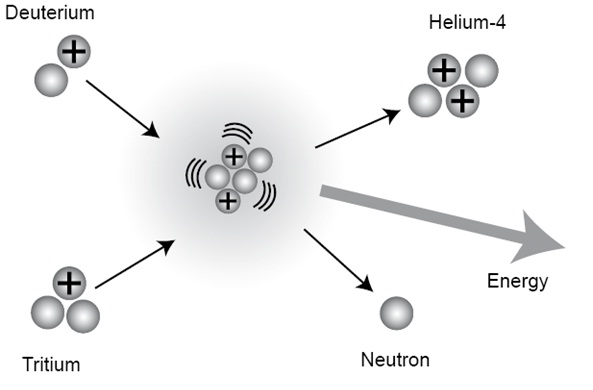
Introduction to Fission & Fusion Nuclear reactions
| 6_fission___fussion.pptx | |
| File Size: | 2209 kb |
| File Type: | pptx |