Learning Objectives
- Explain ionisation and give examples
- Describe the difference between absorption spectrum and an emission spectrum
- Use a diffraction grating and spectrometer to observe the emission line spectra of various gas discharge tubes
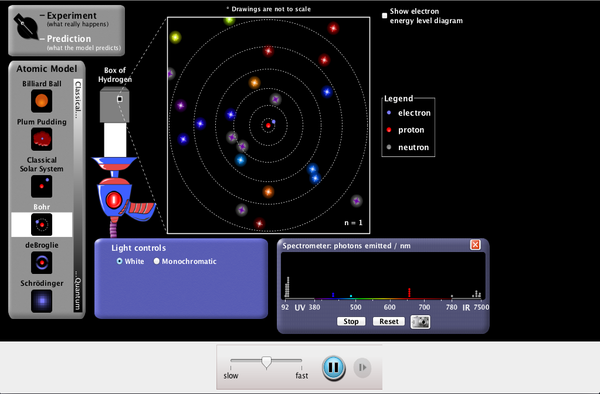
- Explain using Bohr's model for the atom the discrete nature of the spectral lines of the hydrogen atom using equations 1/ landa = R(1/s^2- 1/L^2) and the terms Balmer, Lyman and Paschen series
- Understand the concept that electrons in atoms may drop down an energy level and in so doing give off a photon of specific energy equal to the energy level difference, hence En = -hcR/n^2
| |||||||||||||||||||
|
|
|
|
|
|
|
|